Projects Funded
Brain Response to Immune Challenge
Principal Investigator: Jarred Younger, Ph.D.
University of Alabama at Birmingham
Partial project findings published; see Study Update.
The microglia are immune cells in the brain that work like a protective shield to fend off threats, such as infections. In fibromyalgia patients, a significant portion of these cells are stuck in an activated state of “high alert,” but no one knows why.
“The key piece for understanding fibromyalgia is the concept of sensitized or “primed” microglia, which are operating on a hair-trigger,” says Jarred Younger, Ph.D. “Primed microglia react with full force to events that would not normally activate them.” If these cells are on edge, tiny daily triggers could send them into a frazzled state. This may also explain why infections that the body normally tosses to the curb makes the disease worse.
Ordinarily the microglia sit quietly on the sidelines, surveying the environment within the brain. When an infectious agent invades the body, the microglia rapidly transition to an activated state to neutralize the threat. They do so by producing inflammatory substances (called cytokines) and chemical toxins that temporarily harm the neurons. These substances cause flu-like symptoms, similar to fibromyalgia.
Persistently active microglia could overwhelm the energy demands of the brain and cause metabolites to stockpile. So, Younger is using a brain imaging technique to measure chemicals known to be linked to increased microglia activity. He is also assessing the metabolic health of the brain by measuring lactate and brain temperature. Microglia activation consumes more glucose, produces more lactate, and increases brain temperature. It also makes fibromyalgia symptoms worse (see 2019 AFSA-funded study).
Study Design
This study looks at the brain before and after subjects are injected with a tiny amount of lipopolysaccharide (LPS) – an immune challenge. LPS is an endotoxin that tricks the body into thinking there is an infection. This ought to get the microglia’s attention. However, Younger is using a very low dose that should only produce mild symptoms (if any). The goal is to see if a subtle immune challenge produces an exaggerated microglia response in fibromyalgia patients.
While LPS mimics a bacterial invasion, it is unclear how the microglia in the brain get wind of it. After all, LPS does not normally cross the blood-brain barrier (at least not in healthy people). There must be substances in the blood sending exaggerated distress alarms to the brain. Younger suspects an appetite-regulating hormone called leptin is a likely culprit.
Why leptin? For starters, blood levels of this hormone correlate with body pain in otherwise healthy women. In fibromyalgia patients, leptin levels are twice that of normal. But there is more. Leptin readily crosses from the blood into the brain, so it can directly influence the microglia.
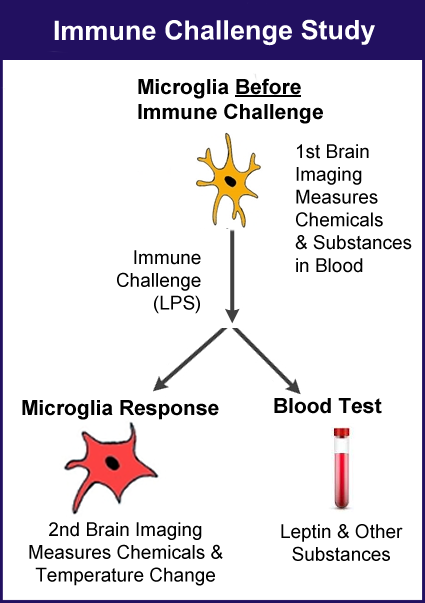
Younger will use molecular resonance spectroscopic (MRS) imaging of the brain to measure various metabolic products and the temperature throughout the brain. Leptin and other chemicals in the blood will be assessed as well (before and after LPS challenge), along with symptoms. Twelve fibromyalgia patients will be compared to 13 healthy control subjects.
Younger’s study involves a three-step process:
- Sample blood and perform the first brain scan.
- Inject all subjects with LPS and wait two hours.
- Sample blood and perform a second brain scan.
Study Update
All study data is analyzed, but only the brain imaging data after the immune challenge is published. This update offers a comprehensive picture of the project (details on what’s been published and general commentary on to-be-published data). One of the study goals was to provide a snapshot of the brain and blood under normal, daily conditions (Baseline Findings). A second goal was to show what occurs in the brain and the blood after subjects are injected with LPS (After Immune Challenge). So, the results are split into two sections: Baseline and After Immune Challenge.
Baseline Findings
“We found very clear baseline differences between people with fibromyalgia and healthy controls,” says Younger. The details are being prepared for a separate report that will be published soon. We will report on them once they are available.
A key marker of microglia activation, which leads to inflammation, is elevated in the brains of fibromyalgia patients compared to the healthy subjects. The impact microglia activation has on the brain’s chemical environment will be published in Younger’s second report. Keep in mind, this data is before the immune challenge.
What about lactate levels? “Lactate should be low in the brain at all times,” says Younger. “You really only see lactate elevations if the brain runs out of glucose.” When microglia are in the activated and inflammatory state, they consume a lot of glucose. So, the accumulation of lactate can represent another sign of brain inflammation.
After Immune Challenge
The Brain
The fibromyalgia group did experience mild flu-like symptoms after injection of the LPS (the immune challenge), but they were gone within 24 hours.* This was surprising because the dose used was substantially less than any other studies involving this same type of immune challenge. However, it shows that fibromyalgia patients are extremely sensitive to very subtle immune challenges, and this is documented in the first published report for this project.
Brain temperature significantly increased in the fibromyalgia group (roughly 1° F), but it did not change in the healthy group. “The most significant group difference in brain temperature was in the right thalamus,” says Younger. “A higher temperature in this area may indicate an abnormally increased inflammatory response.”
If the microglia are on the rampage, how are the brain’s neurons holding up? After all, when the microglia are activated, they become larger and pour out lots of substances into the area. Younger found that the concentration of neurons in several brain regions was decreased by the immune challenge. Chances are that the neurons simply become outnumbered by the “plumped up” microglia in the inflammatory state.
The Blood
Brain imaging provides a window into the brain, but it’s expensive. That’s why Younger ran an elaborate blood panel on all subjects (before and after LPS) to determine if changes in the brain corresponded with alterations in the blood. This portion of the study will appear in a separate publication. However, a prior study by Younger indicates what happens in the blood of fibromyalgia patients following an immune challenge with LPS. But brain imaging was not part of this older study, which is why data from the AFSA-funded project is essential.
What about changes in the blood after an immune challenge with LPS? “The fibromyalgia group had a drastic increase in leptin that was not seen in the healthy controls,” says Younger in a separate publication.** Leptin is a hormone involved in appetite regulation, but it also sensitizes the microglia and makes them secrete inflammatory substances. In fact, elevations of several inflammatory cytokine chemicals were found.
“Our study suggests that abnormal leptin levels in fibromyalgia may be particularly pronounced after an immune trigger,” says Younger. Indeed, he recommends that leptin be further explored as a factor that generates the pain, fatigue, and other symptoms of fibromyalgia.
Ordinarily, leptin suppresses appetite, so why do many fibromyalgia patients struggle with weight gain? One explanation relates to a concept called “leptin resistance.” The baseline levels of leptin are twice that of normal in people with fibromyalgia and they surge to higher levels whenever the body is challenged. Over time, the brain may not respond to leptin as it should. Appetite is not suppressed, and the fat cells produce even more leptin, causing additional pain and weight gain.
Another finding by Younger’s older report: blood glucose levels are significantly less in the fibromyalgia group compared to the controls. Any type of challenge (immune or stress) should cause a surge in glucose levels to supply the muscles, brain, and organs with fuel. Reduced glucose response impedes muscle function (causing pain and fatigue). It also short-changes the brain, contributing to mental fatigue, fibrofog, and other symptoms.
As soon as Younger publishes the baseline brain data and the corresponding blood assays, the information will be posted here.
* Mueller C, Younger JW, et al. Abnormal immune system response in the brain of women with Fibromyalgia after experimental endotoxin challenge. Brain Behav Immun Health 30 (2023):100624. Free AFSA-funded report.
** Jones C, Younger J, et al. Altered response to Toll-like receptor 4 activation in fibromyalgia: A low-dose, human experimental endotoxemia pilot study. Brain Behav Immun Health 34 (2023):100707. Free report.
